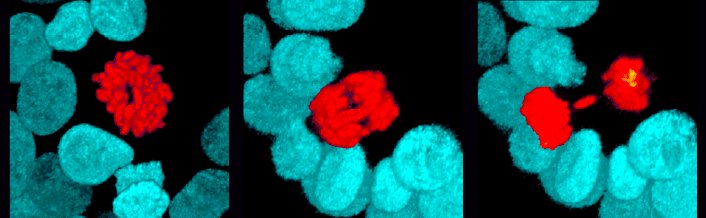
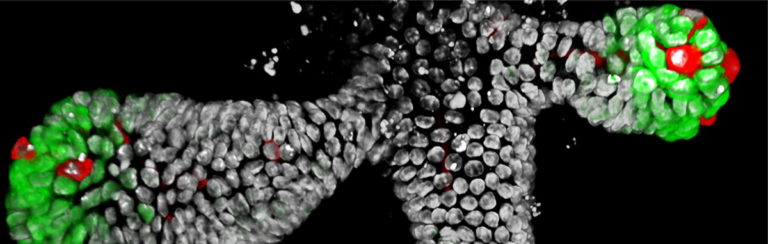
Joris Hageman successfully defended his thesis ‘Phenotypic plasticity of human intestinal stem cells in regeneration and cancer’. He made such a great show of his defense, that he was awarded cum laude. Wow!!! Joris was our best in shaping beautiful looking Bonsai-organoids. We wish him all the best in his further career and life.